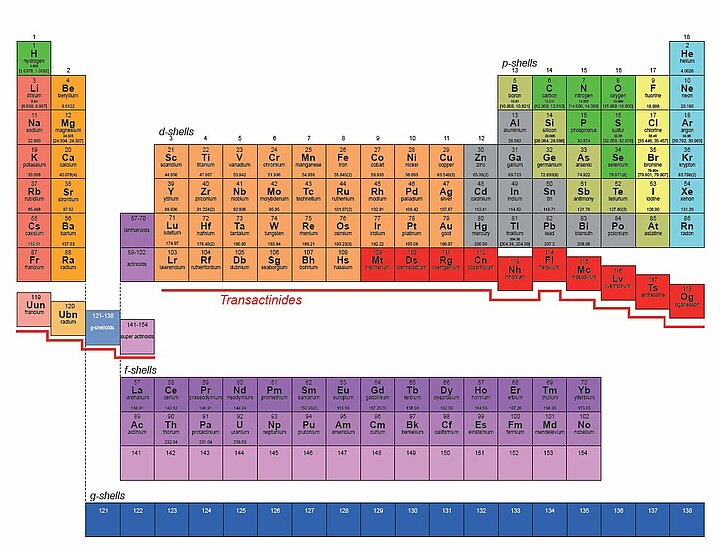
The Mendeleev table is currently composed of 118 chemical elements. Benoît Gall travelled to Russia and Japan in search of elements 119 and 120, that have never yet been observed. Gall, a researcher from the Hubert Curien Pluridisciplinary Institute, tells us about his quest for the Grail.
It took nine years and three full years of beam time for the Japanese to discover nihonium, the chemical element of atomic number 113
, says Benoît Gall, as he talks of a challenge that was both passionate and frustrating and for which patience is a key factor. He has just returned from a two-week trip to Russia where, for over 20 years, he has been working with the FLNR laboratory at Dubna, 100 km north of Moscow.
At first, his work there concentrated on the spectroscopy of heavy elements, but recently the Russian laboratory has acquired the SHE Factory, namely the “superheavy elements” factory. The aim is to study superheavy nuclei (Z≥104) and be able to observe new chemical elements, including the famous element 120. In order to do this, the researchers use the fusion of two “target” and “beam” atoms.
“The probability of survival is extremely weak”
In order to secure element 120, we have chosen to use a titanium-50 beam (Z=22) and a californium-249 target (Z=98).
By adding together other elements, it is also possible to combine 120 protons, but why choose these two? The probability of survival is extremely weak, neutrons and protons have to be transported while limiting the excitation energy of the compound formed. There are only a few rare elements, of which titanium-50 is one, that benefit from excess binding brought about by quantum effects and which allow the reaction to stabilise. It is therefore the element for which the probability of synthesis is the greatest.
The next thing is to get hold of them. The IPHC, which alone masters the production of these rare volatile compounds, is charged with creating isotopic titanium organo-metallic molecules. As regards californium-249, this should be provided by the American Oak Ridge National Laboratory, provided that diplomatic relations between the two countries allow for it. If not, we could also use a Russian curium-248 target (Z=96) with a chrome-54 beam (Z=24), which is also produced by the IPHC. But the reaction would be three to ten times less efficient.
The first titanium and chrome beam tests have recently been accelerated by the SHE Factory and adjustments will follow in the next fortnight to reach the awaited intensities. Within the next month, and if the health crisis does not prevent him from doing so, Benoît Gall should be returning to Russia for the first target tests.
Superheavy elements with unknown properties
These are radioactive elements. They must first survive long enough to be observed…. at the moment they disappear!
emphasises Gall, who explains that because of this short lifespan, industrial applications are not possible. Once their existence is established, it is however possible to test their chemical properties.
In parallel with this research, Benoît Gall and his team also collaborate with the Japanese to find element 119. These are the last elements that we will be able to identify before no longer being able to push the technological limits. We still hope to find 121 and 122 in the next 10-15 years; we would then be on the g-shell of the periodic table, i.e. where elements of unknown properties reside, provided that the chemistry envisaged here, due to the influence of relativistic effects, is correct… but precisely due to these relativistic effects, the periodic table may need some adjustments and one may already have some effect of the g-shell before Z=121. This is exactly why a machine like the SHE-factory, able to produce dozens of these rare elements, was built.
Marion Riegert
More information
Benoît Gall has just won a USIAS fellowship to work on targets capable of withstanding these unprecedented beam intensities and to develop new, more resistant materials. Often, the beams are too strong and the target breaks
, Gall explains.